CorDx Influenza Test A + B, COVID-19, RSV, Combi Test 4in1 is an antigen test. Covid, influenza A/B and RSV can be detected within 15 minutes using a simple nasal swab.
€6.51
Content:
1 Stück
(May vary by country)
Ready to ship in 1-3 working days ⁴
Product number:
RA5731814
Stück:
Manufacturer:
Porod Medizintechnik GmbH
Horner Straße 24, 3580 Frauenhofen, Österreich
Product information "CorDx influenza test A + B, COVID-19, RSV, combination test 4in1"
CorDx Influenza Test A + B, COVID-19, RSV, Combi Test 4in1 is an antigen test. Covid, influenza A/B and RSV can be detected within 15 minutes using a simple nasal swab.
The Influenza A, Influenza B, COVID-19 and RSV (Respiratory Syncytial Virus) Combitest is an immunochromatographic in vitro assay for the qualitative and differential detection of nucleocapsid protein antigens of influenza A (including subtype H1N1), influenza B, respiratory syncytial virus and/or SARS-CoV-2 in nasal swab specimens from individuals with or without symptoms or other epidemiological reasons for suspecting the presence of influenza A, influenza B, RSV and/or COVID-19 infections. This Covid Influenza RSV combination test is for in vitro diagnostics and self-testing.
Advantages of the CorDx Covid Influenza RSV combination test
The CorDx Covid Influenza RSV Combo Test is a qualitative test that is able to detect Covid-19, RSV, influenza A and B with a high sensitivity of over 96% and a specificity of over 99%. It is quick and easy to perform and is suitable for both employees and private individuals seeking certainty in the event of illness.
- Quick to perform: The test can be carried out in just a few minutes.
- 4in1 combination test: Covid, influenza A/B and RSV
- Evaluation after 15 minutes
- Easy to use: The test is simple to perform, just swab your nose.
- Storage at 2 - 30 °C CE certified clinically tested
- Includes all necessary utensils
Clinical performance
The clinical performance characteristics of the CorDx Influenza A/B, COVID-19, RSV Combo Ag test were evaluated in the clinical studies. A total of 452 symptomatic or asymptomatic subjects were recruited for the clinical study of the Influenza A/B and RSV test. In addition, a study was conducted with 560 symptomatic or asymptomatic suspects of COVID-19.
The test results are as follows:
- Influenza A: For influenza A antigen detection, the positive coincidence rate is 100.00%, the negative coincidence rate is 99.34% and the overall coincidence rate is 99.43%.
- Influenza B: When detecting the influenza B antigen, the positive coincidence rate is 96.00 %, the negative coincidence rate is 99.67 % and the overall coincidence rate is 99.15 %.
- RSV: For RSV antigen detection, the positive coincidence rate is 98.98%, the negative coincidence rate is 99.21% and the overall coincidence rate is 99.14%.
- Covid-19: For COVID-19 antigen detection, the positive coincidence rate is 89.09%, i.e. 98.67% for Ct≤25, 93.00% for Ct≤30, and the negative coincidence rate is 100.00%, the overall coincidence rate is 97.86%.
Detection limit (analytical sensitivity)
- Influenza A: 1.5×104 TCID50/mL
- Influenza B: 1.5×105 TCID50/mL
- Covid-19: 200 TCID50/mL
- RSV: 1.0×104 TCID50/mL
Test principle of the CorDx Influenza Test A + B, COVID-19, RSV, 4in1 self-test combination test
Influenza A and influenza B
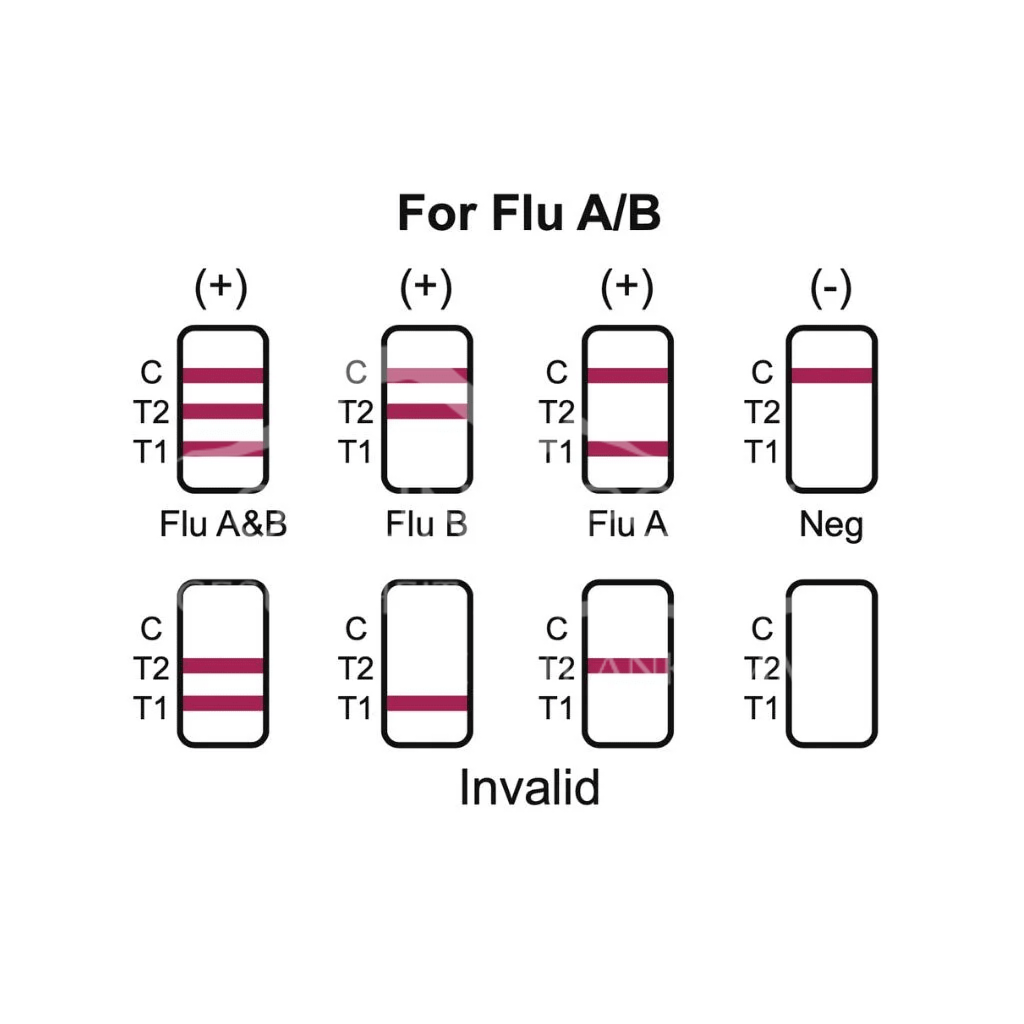
- The Influenza A, Influenza B Antigen Test Strip uses monoclonal mouse anti-influenza A antibodies (T1), monoclonal mouse anti-influenza B antibodies (T2) and polyclonal goat anti-mouse IgG antibodies (C), each immobilised on a nitrocellulose membrane. The test uses colloidal gold to label monoclonal mouse anti-influenza A antibodies and monoclonal mouse anti-influenza B antibodies. Nanocolloidal gold technology is used and highly specific antibody-antigen reactions and the principle of immunochromatographic analysis technology are applied. During testing, the influenza A antigens in the sample combine with the colloidal gold-labelled mouse anti-influenza A monoclonal antibody to form a complex, which then combines with the mouse anti-influenza A monoclonal antibody coated in the T1 test line during chromatography to form a red line in the T1 line.
- During testing, the influenza B antigens in the sample combine with the colloidal gold-labelled mouse anti-influenza B monoclonal antibody to form a complex, which then combines with the mouse anti-influenza B monoclonal antibody coated in the T2 test line during chromatography to form a red line in the T2 line. If the samples do not contain influenza type A and B antigens, no red lines will appear in the T1 and T2 areas. Regardless of the presence of influenza type A or B antigens in the sample, a red line will always appear in the quality control area (C) if possible. The red line in the quality control area (C) is used to check whether 1. a sufficient sample volume has been added, 2. a proper flow rate has been achieved, 3. and as a quality control for the reagents.
Covid-19
- The COVID-19, RSV Antigen Test Strip uses mouse monoclonal anti-COVID-19 antibody (T2), mouse monoclonal anti-RSV antibody (T1) and goat polyclonal anti-mouse IgG antibody (C), each immobilised on a nitrocellulose membrane. It uses colloidal gold to label the mouse monoclonal anti-COVID-19 antibody and the mouse monoclonal anti-RSV antibody. The nanocolloidal gold technology is used and highly specific antibody-antigen reactions and the principle of immunochromatographic analysis technology are applied. During testing, the COVID-19 antigen in the sample combines with the colloidal gold-labelled mouse monoclonal anti-COVID-19 antibody to form a complex, which was then combined with the mouse monoclonal anti-COVID-19 antibody coated in the T2 test line during chromatography; at this point, there is a red line in the T2 area (the test is positive).
RSV
- The RSV antigen in the sample combined with the RSV monoclonal antibody labelled with colloidal gold to form a complex, which was then combined with the mouse monoclonal anti-RSV antibody coated in the T1 test line during chromatography; at this time, there is a red line in the T1 area (The test is positive). If the samples do not contain COVID-19 and RSV antigens, there are no red lines in the T1 and T2 areas (the test is negative). Regardless of the presence of COVID-19 or RSV virus antigens in the sample, a red line always forms in the quality control area (C). The red line in the quality control area (C) is used to check whether 1. a sufficient sample volume has been added, 2. a proper flow rate has been achieved, and 3. as a quality control for the reagents. If this does not occur, the test must not be analysed.
General information on the CorDx Influenza Test A + B, COVID-19, RSV, Combi Test 4in1 self-test
The test is intended to help with the rapid diagnosis of infections with influenza A, influenza B, respiratory syncytial virus and/or SARS-CoV-2. This test is intended for non-prescription home use with self-collected nasal swabs from individuals 14 years of age and older with symptoms of influenza A, influenza B, RSV and/or COVID-19 within the first 7 days of symptom onset. This test is also intended for use with adult nasal swabs collected from individuals 2 years of age and older with symptoms of influenza A, influenza B, RSV and/or COVID-19 within the first 7 days of symptom onset. More false-positive results may occur in people without COVID-19 symptoms and/or people living in areas with a low number of COVID-19 infections and no known exposure to COVID-19. Persons without symptoms should only be tested if they are contacts of confirmed or probable cases or if there are other epidemiological reasons to suspect influenza A/B or RSV/COVID-19 infection. An additional confirmatory test should also be carried out using a molecular test (e.g. PCR).
This test only provides preliminary test results. Therefore, all positive results of this combination antigen test for influenza A, influenza B, COVID-19 and/or RSV must be confirmed by alternative test methods and clinical findings.
Dosage form
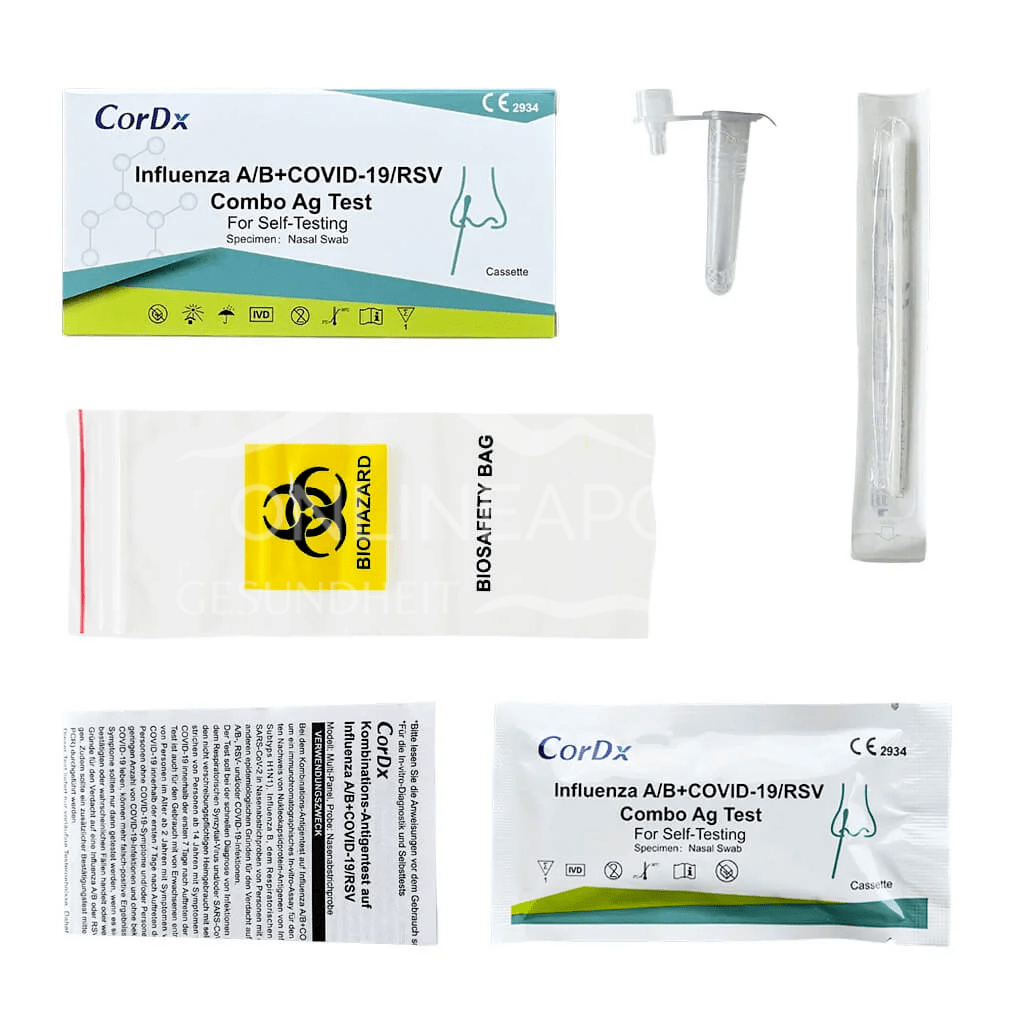
The test kit of the combination antigen test for influenza A/B, COVID-19, RSV contains the following components for performing the test: 1 test cassette, 1 instruction for use, 1 sample collection tube with sample processing solution, 1 swab for sample collection, 1 tube stand, 1 collection bag.
Application
Please see package insert.
| Piece: | 1 |
|---|
Read more
No reviews found. Share your insights with others.
Warnings - CorDx influenza test A + B, COVID-19, RSV, combination test 4in1
Instructions for use
Product recommendations for you