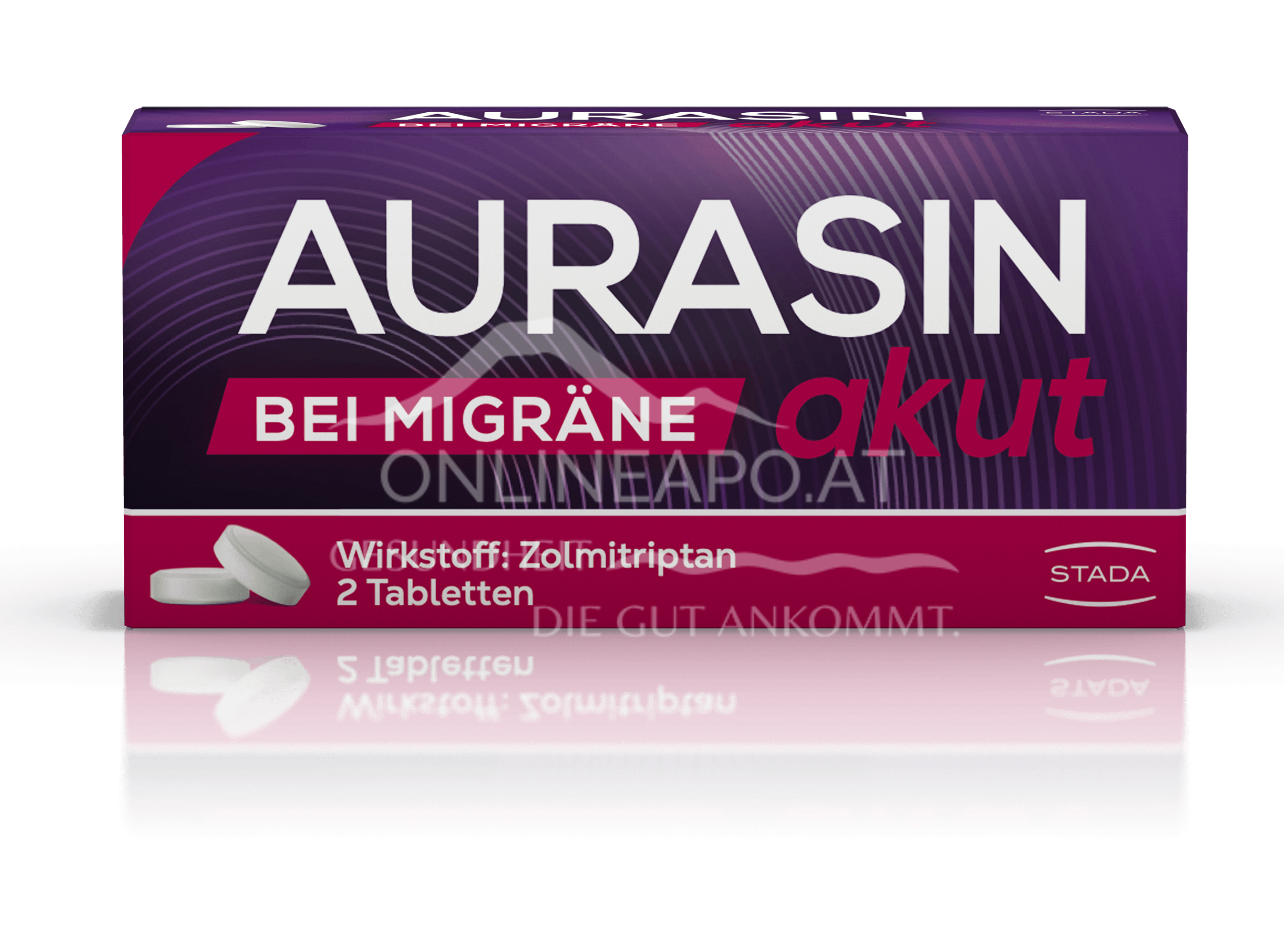
Naproxen sodium, the active ingredient in Aleve - film-coated tablets, belongs to the group of non-steroidal anti-inflammatory drugs (NSAIDs) that have analgesic, antipyretic and anti-inflammatory effects.
€13.65
(May vary by country)
Available, delivery time: 1-3 days ⁴
Naproxen sodium, the active ingredient in Aleve film-coated tablets, belongs to the group of non-steroidal (= "cortisone-free") medicines (NSAIDs) that have analgesic, antipyretic and anti-inflammatory effects.
For the relief of mild to moderate pain such as
- Headache
- Toothache
- Back pain
- Menstrual cramps
- Pain associated with colds
Pain in the gastrointestinal tract must not be treated with naproxen.
Properties
- Contains 200 mg naproxen
- For adults and adolescents from the age of 12
- Analgesic, antipyretic and anti-inflammatory
Dosage form
Film-coated tablets
Intended use
- Adolescents aged 12 years and over and weighing at least 50 kg and adults: 1 film-coated tablet every 8 - 12 hours. The daily dose of 3 film-coated tablets must not be exceeded without medical advice.
- Children up to 12 years of age and persons under 50 kg body weight: Due to the high active substance content, Aleve film-coated tablets are not suitable for use in these patient groups, as the dosage cannot be adjusted according to body weight.
- Route of administration: Oral use. Take Aleve film-coated tablets whole with plenty of liquid at mealtimes. Aleve - film-coated tablets are more effective when taken on an empty stomach. For patients with a sensitive stomach, it is advisable to take Aleve film-coated tablets during meals.
- Duration of use: Aleve - film-coated tablets must not be taken for longer than 4 days without medical advice. If there is no improvement in the first 4 days, a doctor should also be consulted.
Ingredients
- The active substance is: Naproxen.
- 1 film-coated tablet contains 220 mg naproxen sodium (equivalent to 200 mg naproxen).
- The other ingredients are Tablet core: microcrystalline cellulose, magnesium stearate, povidone K 30, talc. Tablet coating: colouring agents (E 132, E 171).
| Alcohol-free: | Not specified |
|---|---|
| Gluten-free: | Not specified |
| Lactose-free: | Not specified |
| Perfume: | Not specified |
| Piece: | 24 |
| Sugar-free: | Not specified |
| Vegan: | Not specified |
Instructions for use
| Type of use: | Unzerkaut mit reichlich Flüssigkeit zu den Mahlzeiten einnehmen |
|---|---|
| Field of use: | Symptomatisch bei leichten bis mittelschweren Schmerzen |
| Dosage: | Personen ab 12 Jahren mind 50 kg KG 1 FT alle 8 bis 12 Std; MTD 3 FT Ohne ärztlichen Rat max 4 Tage anwenden Niereninsuffizienz Dosisreduktion bei CrCl 30mlmin Nicht anwenden bei CrCl 30 mlmin Nicht für Kinder unter 12 Jahren oder |
| Properties and efficacy: | Nichsteroidaler Entzündungshemmer |
| Contraindications: | LogImport.sisx.GAD.Analgetika-Asthma, Bluterkrankungen, schwere Herzinsuffizienz, MagenDarm-Ulzera, Blutungen, schwere Leber- oder Niereninsuffizienz, Kinder bis zum 12 Lebensjahr und Personen unter 50kg KG |
| Side effects: | MagenDarm, Leber, Psyche, ZNS, Ödeme, Bluthochdruck, Herzinsuffizienz, schwere Hautreaktionen, Überempfindlichkeit, Niere, Labor, Kouinis-Syndrom |
| Special warnings for safe use: | Vorsicht bei Hypertonie, Gefäßerkrankungen, Autoimmunerkrankungen, Porphyrie Auf MagenDarm-Blutungen, Ulzera, Perforationen, schwere Hautreaktionen achten |
| Interactions: | Vorsicht Andere Entzündungshemmer erhöhtes Nebenwirkungsrisiko, Herzglykoside, Gerinnungshemmer, Lithium, Phenytoin, Diuretika, Blutdrucksenker abgeschwächt, Methotrexat, Immunsuppressiva, orale Antidiabetika, Alkohol Gerinnungshemme |
| Pregnancy and breastfeeding period: | Zwingende Indikationsstellung im 1 und 2 Trimenon, kontraindiziert im 3 Trimenon und in der Stillzeit |
Pregnancy information
| Use forbidden |
|---|
| Use only if absolutely or vitally indicated |
Breastfeeding information
| Breastfeeding forbidden |
|---|
Pictograms
| Caution photosensitivity, avoid excessive UV exposure |
|---|
| Take with meals |
| Caution for diabetics |
| Take intact |
| Caution: may affect ability to drive/use machines |